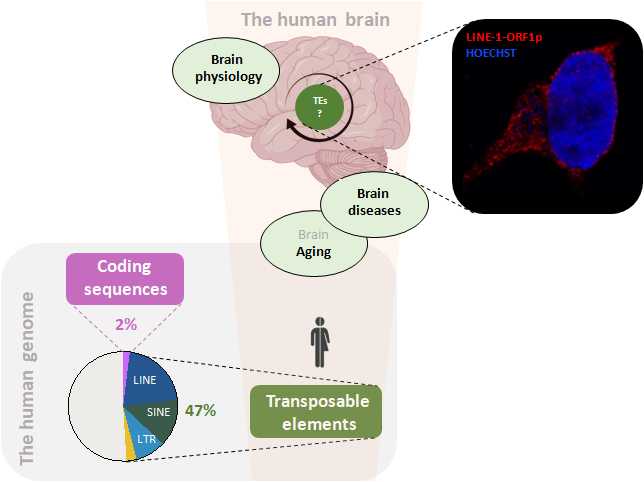
Our team is interested in understanding the role of transposable elements (TEs) in the physiology and pathophysiology of the brain. TEs comprise the “dark side” of the genome of many organisms including humans and have gained growing interest as drivers of evolution but also as genomic elements associated with cancer, auto-immune and neurological diseases. We find the retrotransposon LINE-1 encoded protein ORF1p is expressed at steady-state in the mouse and human brain. Yet not much is known about the regulation and expression patterns of LINE-1 in the brain nor how this expression might affect brain physiology or pathophysiology. Recent evidence from our group suggests that an uncontrolled activation of LINE-1, normally repressed by various cellular levels of control including at the chromatin level, is at the origin of genomic instability in and stress-triggered degeneration of dopaminergic neurons (Blaudin de Thé et al, EMBO J, 2018; Ravel-Godreuil et al, iScience, 2021). However, it is currently not known to what extend and how the expression and activity of TEs is associated with and causally connected to neurodegenerative diseases (NDs). We currently investigate the hypothesis that chromatin decompaction triggered by brain aging or genetic factors leads to the de-repression of TEs which can be neurotoxic by inducing genomic instability and neuroinflammation. To study the cellular and molecular mechanisms that link TEs to neuronal physiology and pathophysiology, we use human, mouse and primate neuronal cells and brain tissues and, besides a wide-range of standard biochemical, cellular and molecular biological techniques, also next-generation sequencing and bioinformatics, proteomics, fluorescent-activated nuclei sorting and state-of-the-art imaging. Understanding the role of TEs in NDs will increase our understanding of the pathogenesis of Parkinson’s disease (PD) and Alzheimer’s disease (AD) but will also provide a new and original framework for the development of disease-modifying treatments.