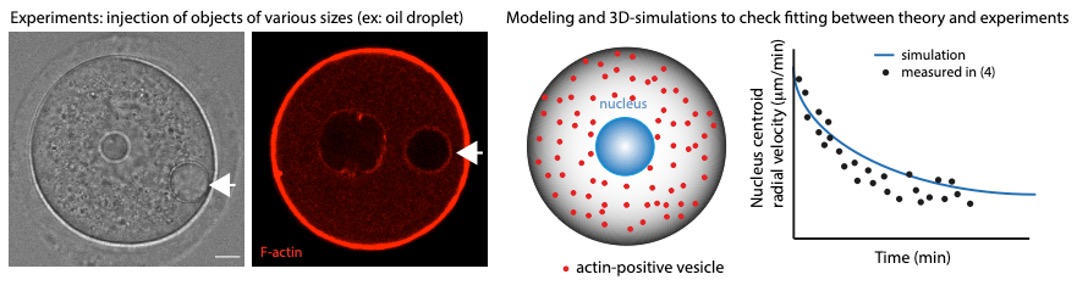
Our goal aims at understanding how a female gamete turns into a viable embryo, using morphological and biophysical approaches of meiotic divisions.
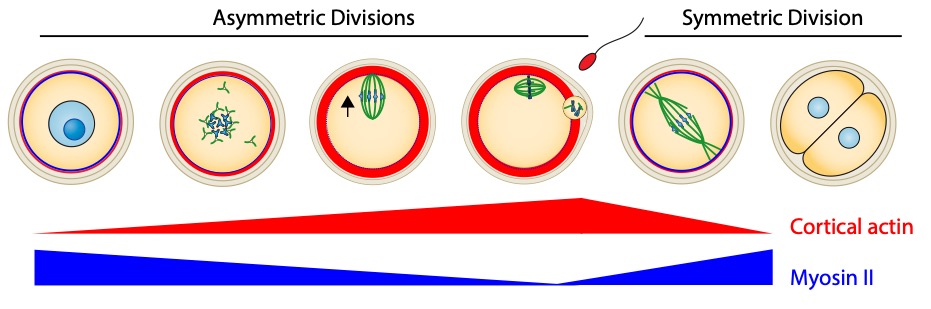
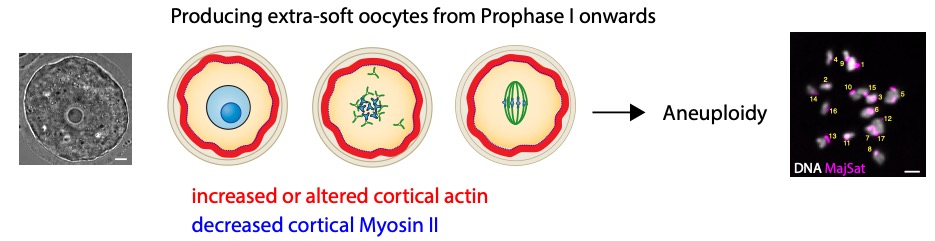
Organisms that propagate via sexual reproduction often come from the fusion between an oocyte and a sperm. While oocytes are transmitted in full to the next generation, the sperm mostly transmit their genomes. We study how this maternal inheritance is preserved and what are the consequences when it goes awry. For this, we study the last stages of murine oogenesis, namely oocyte end of growth as well as meiotic divisions,which are extremely asymmetric in size of the daughter cells, allowing to preservethe maternal stores accumulated during growthto support early embryo development (Fig 1). Oocytes have to accomplish two opposite tasks: segregate their chromosomes equally while partitioning their cytoplasm unequally. This tour de force is challenged by the lack of canonical centres of microtubule nucleation, namely centrosomes containing a pair of centrioles. Centrosomes of mitotic cells organize spindle as well as astral microtubules at spindle poles, mediating spindle morphogenesis and positioning. Over the past years, we have discoveredthe unprecedented impact of actin-mediated mechanisms regulating meiotic chromosome position on gamete fitness.
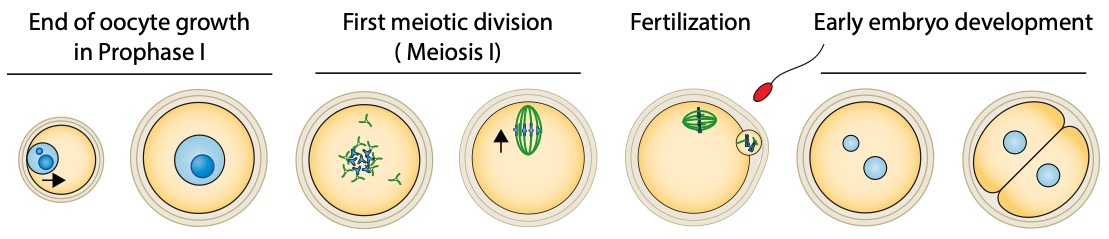
During their growth in the ovary, oocytes are arrested in Prophase I of meiosis. The oocyte nucleus is centralized at the end of growth. They physiologically and cyclically resume meiosis and undergo two successive asymmetric meiotic divisions, without intervening DNA replication. Nuclear envelope breakdown and spindle assembly mark the entry into meiotic divisions. DNA in blue (dark bluefor nucleolus) and microtubules in green. The black arrows indicate the global direction of chromosome motion.