Abstract
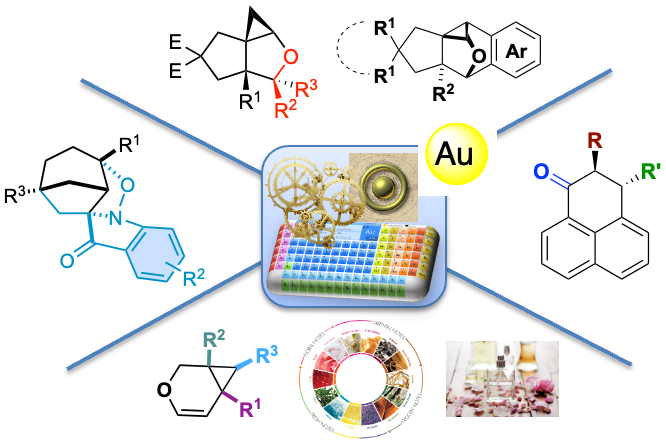
Over the past 20 years, significant research has been directed toward the development of new methodologies for synthetic efficiency and atom economy processes in the presence of gold complexes.1 We have been initially engaged in a wide project dedicated to the development of catalytic methodologies for the synthesis of original and functionalized carbo- and heterocycles.2Our group recently described the gold-catalyzed domino reaction of cycloisomerization/ nucleophilicaddition/C→Orearrangement, leading to a wide range of functionalized polycyclic ketones.3 An efficient 2-step process involving an Au-catalyzed Prins-type cyclization and a Pd-catalyzed cyclopropane C-H activation was also disclosed for the synthesis of polysubstituted cyclopropanes. We have also developed a divergent cascade cycloisomerization / [3 + 2] vs [2+2+1]-cycloaddition via gold or silver catalysis in 1,1,1,3,3,3-hexafluoroisopropan-2-ol (HFIP).4 We prepared low molecular weight enyne derivatives and optimized the reaction conditions allowing functionalized volatile oxa-bicyclo[4.1.0]-hept-4-ene in good to excellent isolated yields. The synthetic interest of these low molecular weight bicyclic enols was further demonstrated by the unprecedented olfactory evaluation of the bicyclic derivatives fragrances.5 Finally, our group has entered in the field Au(I)-Au(III) via electrochemistry methods as well as reactivity for Au(III) complexes.6 This presentation will show the latest results on sustainable access to functionalized racemic and chiral carbo- and heterocycles.