Astrocytes are brain glial cells with a predominant influence on the cerebrovascular system controlling perivascular homeostasis, blood-brain barrier (BBB) integrity, crosstalk with the peripheral immune system, endothelial transport or vessel contractility in response to neuronal activity. Astroglial regulation towards the cerebrovascular system is set at the level of the gliovascular unit (GVU), a specialized interface composed by astrocyte perivascular processes (PvAP) or endfeet that fully sheath the brain vessels composed by endothelial and mural cells (pericytes and vascular smooth muscle cells (VSMC))3. Such morphological and functional polarity is of critical importance for the brain physiology and is altered in several pathological contexts including epilepsy, ischemic brain damage, Alzheimer’s disease or multiple sclerosis. Thus, the way functional polarization is set in astrocytes and the mechanisms by which astrocytes accomplish their regulatory functions at the brain vascular interface are questions of prime importance.
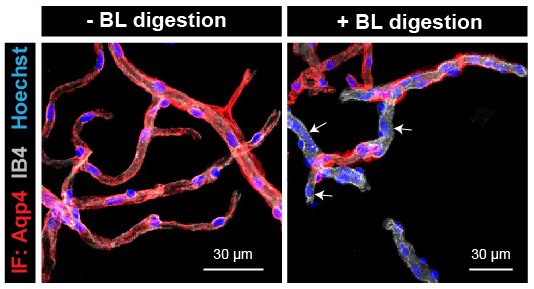
Our group elaborated an innovative and original research program to investigate how astrocytes regulate the physiology and physiopathology of the cerebrovascular system. We developed a toolkit enabling GVU examination: i) the purification of the GVU: we demonstrated that PvAPs separated from their soma remain attached to the surface of mechanically purified brain vessels (Fig.1). We showed that mild enzymatic digestion of the basal lamina led to purified vessels with partially detached PvAPs from the vessel surface. A comparison of digested and non-digested samples allowed us to identify PvAPs components, such as mRNAs transported in PvAPs. These experimental protocols provided an opportunity for the biochemical and molecular characterization of PvAPs; ii) We developed a translating ribosome affinity purification (TRAP) technique based on an astrocyte-specific ribotagging approach (TRAP transgenic mouse strain: Aldh1L1 eGFP Rpl10a) to isolate polysomal mRNAs in astrocytes. To purify ribosome-bound mRNAs in PvAPs or perisynaptic astrocytic processes (PAP), we combined TRAP with the purification of the GVU or synaptogliosomes.
We developed a three-dimensional in situ method, AstroDot and a dedicated bioinformatics tool, AstroStat, for identifying and quantifying mRNA distribution in astrocytes (Fig. 2).
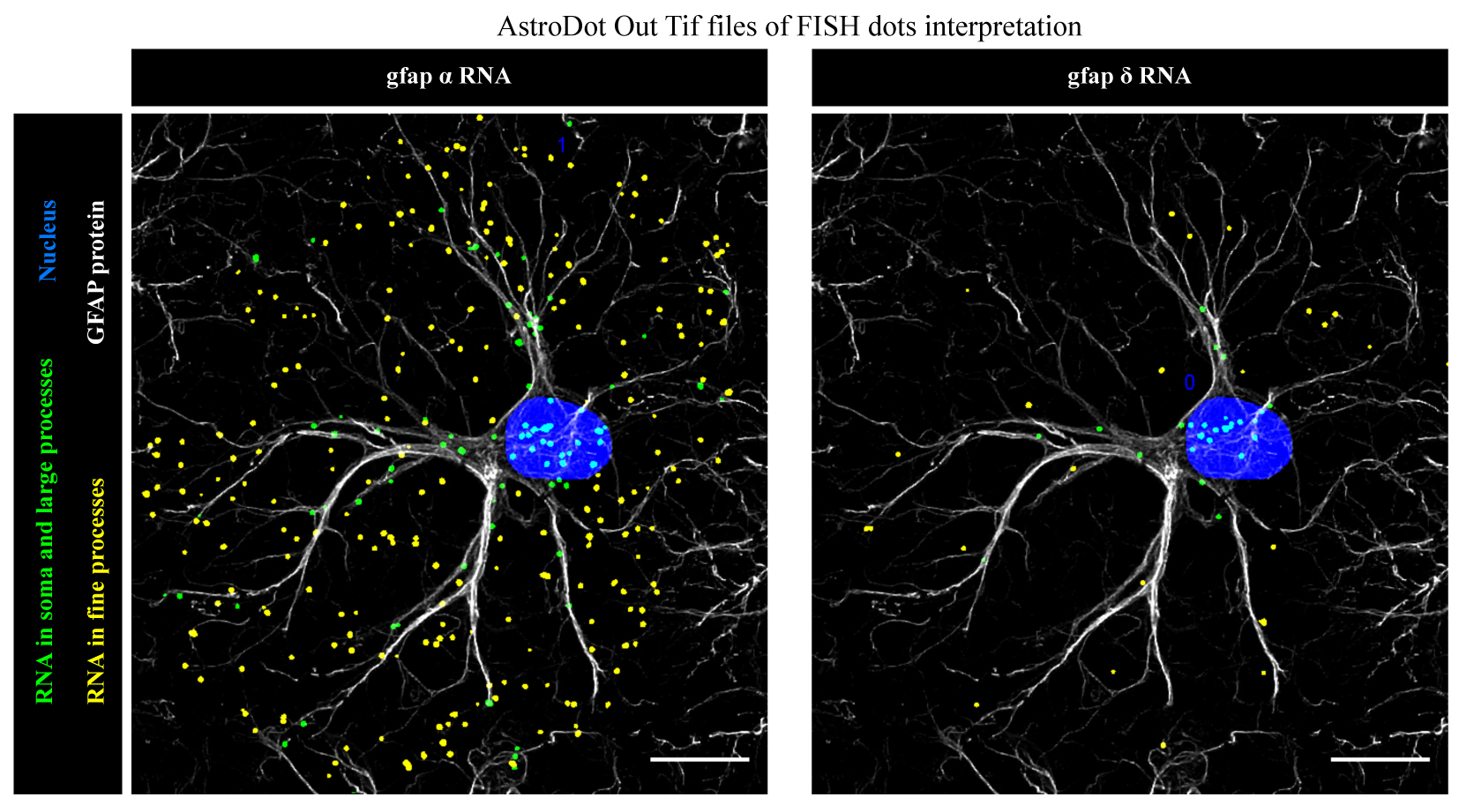
We demonstrated that distal compartmentalization of mRNAs occurs in PvAPs and PAPs, together with translation. We showed that astrocyte processes are equipped with endoplasmic reticulum and Golgi apparatus, suggesting the existence of local routes for protein maturation and trafficking. We characterized the Endfeetome, the astroglial polysomal repertoire of PvAPs in the whole brain, and more recently the Papome, the astroglial polysomal repertoire of PAPs of the dorsal hippocampus. We showed the enrichment of specific pools of polysomal mRNAs in astrocyte interfaces compared to the soma. We identified local translation changes upon fear conditioning in PAPs, suggesting for the first time a physiological link between astrocytic local translation and learning and memory processes. We revealed that mRNAs are redistributed upon astrocyte reactivity in a mouse model of Alzheimer’s disease.
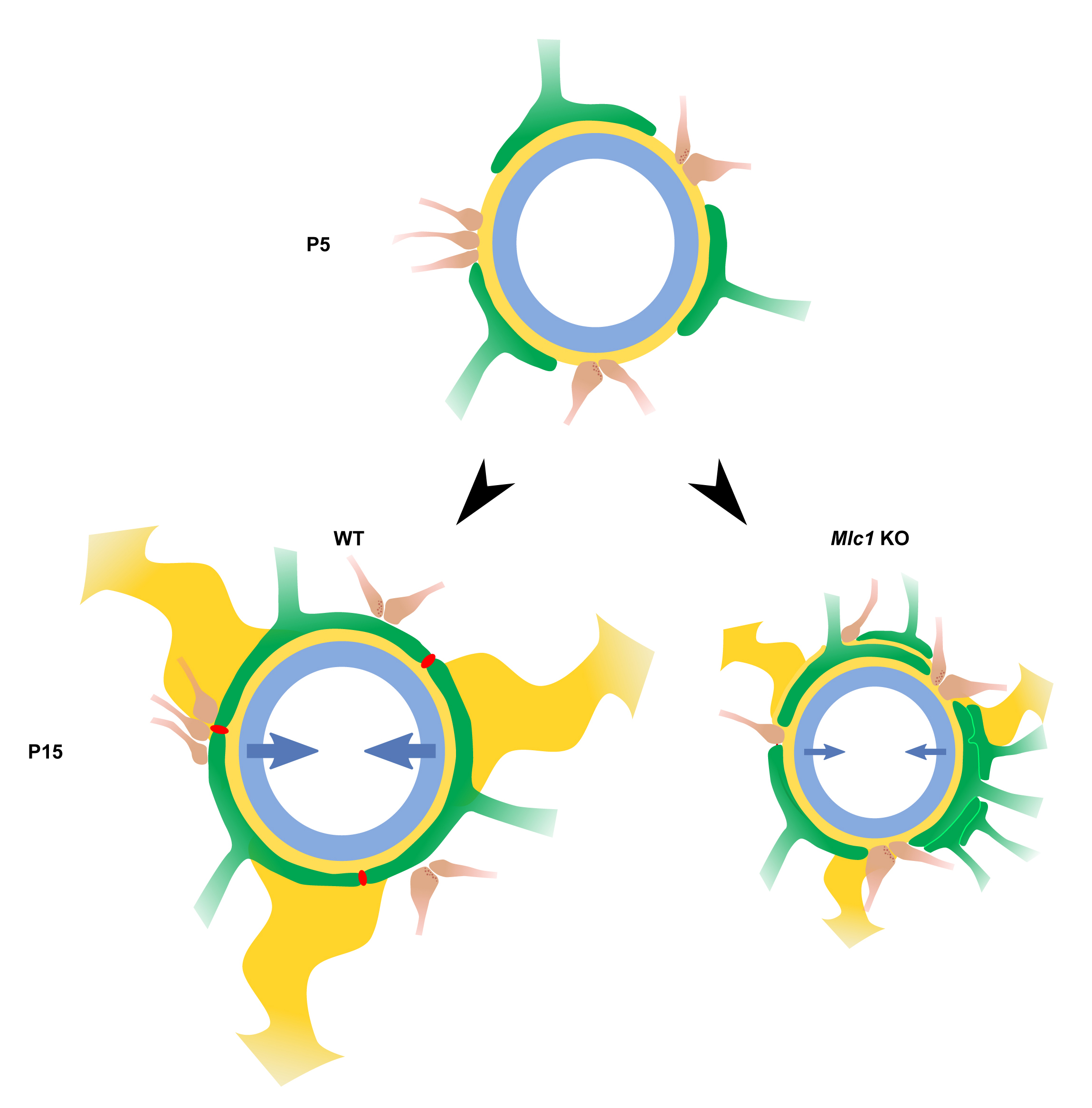
Altogether, these findings conceptually changed the way we think about astrocyte-interfaces signaling. Proteins required for astrocyte functions at their interfaces were thought to be produced and matured in the cell body. Our studies suggested that local translation might be an important mechanism for astrocytic molecular and functional polarity in physiology and pathology. These approaches also provided novel and unexpected insights into the PvAP’s and PAP’s molecular identity, with the preferential translation of some mRNAs in specific astrocyte compartments. We explored the role of several molecules enriched and locally translated in PvAPs and uncovered that: (i) the gap junction protein connexin 43 is central in the regulation of the BBB immune quiescence in the healthy brain and in the context of meningitis; (ii) Dp71, an astrocytic-enriched dystrophin isoform, regulated the perivascular anchorage of the Aquaporin 4 water channel; (iii) the loss of the specific PvAP membrane protein MLC1 caused megalencephalic leukoencephalopathy with subcortical cysts (MLC, a rare type of leukodystrophy) by regulating the development of PvAPs, vascular smooth muscle cells (VSMC) contractility, neurovascular coupling and intraparenchymal fluid circulation (Fig. 3); (iv) the MLC1/GlialCAM complex modulated perivascular GPCR signalization.
Although the contribution of astrocytes in the regulation of the cerebrovascular system is well recognized, how the gliovascular unit develops, is organized and how astrocytes accomplish their functions at the vascular interface is largely unknown. Our ambition is to provide essential knowledge on the mechanisms underlying astroglial control of cerebrovascular functions in health and disease.


