Résumé
Activation of carbene precursors by planar, low-spin cobalt(II) complexes leads to formation of ‘carbene radical’ intermediates.1 These are intrinsically reactive metal-bound carbon radicals (Figure 1), useful for catalytic synthesis of a variety of ring compounds and other useful organic building blocks. These reactions have in common that they proceed via selective radical-type elementary steps, made possible by intra-molecular single-electron transfer from the metal to the substrate.
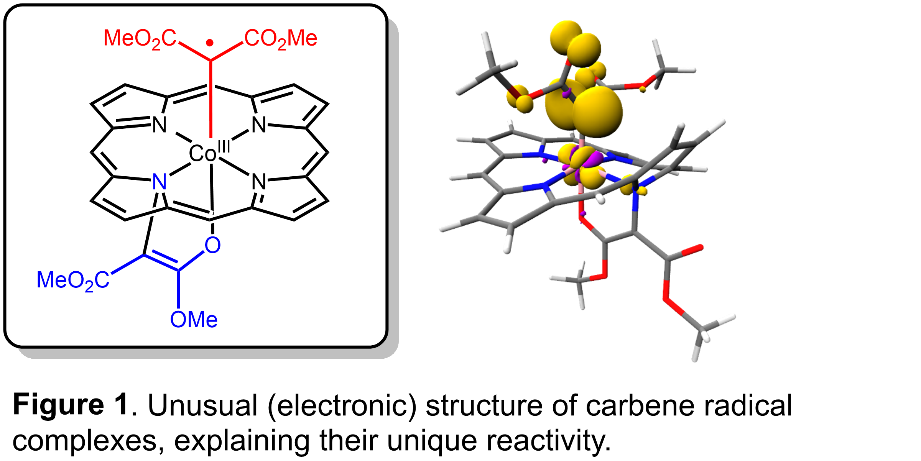
We recently discovered that such carbene radicals are useful for the catalytic synthesis of o‑quidomethide and o-quinodimethane intermediates. These reactive intermediates give access to useful one-pot catalytic procedures to produce e.g. 2H-chromenes[2] and dihydronaphtalenes[3] Similar reactions with nitrene precursors follow related pathways.
In this presentation focus on characterization of the reactive species produced by the intramolecular electron transfer process, and we will demonstrate synthetic applications towards the synthesis of valuable ring compounds, such as dibenzocyclooctenes,[5] monobenzocyclooctadienes,[6] 1H-2-benzoxocins,[7] and pyrrolidines.[8]
